An Immunocompetent Kidney Model to Study Inflammation and Immune-Mediated Injury

Why This Is Important

Challenges

Need
MIMETAS’ Answer
Immunocompetent Proximal Tubule on-a-Chip
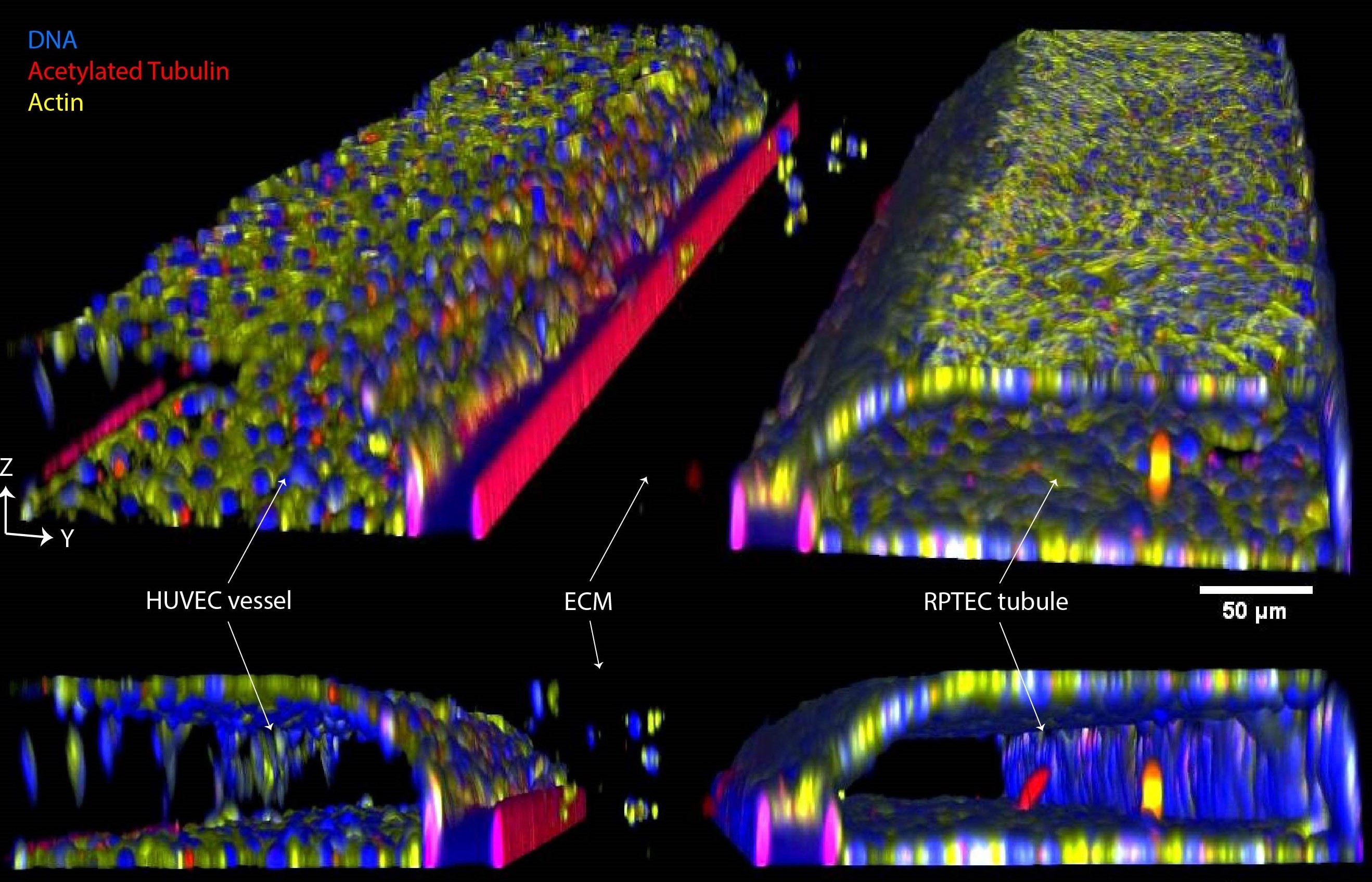
In this model, human renal proximal tubule epithelial cells (RPTECs) are cultured in a first perfusion channel while human endothelial cells (HUVECs) line the second perfusion channel. Both channels are separated by a collagen-I extracellular matrix and cultured under perfusion flow, resulting in theformation of polarized epithelial tubules and a vascular interface (figure 1).
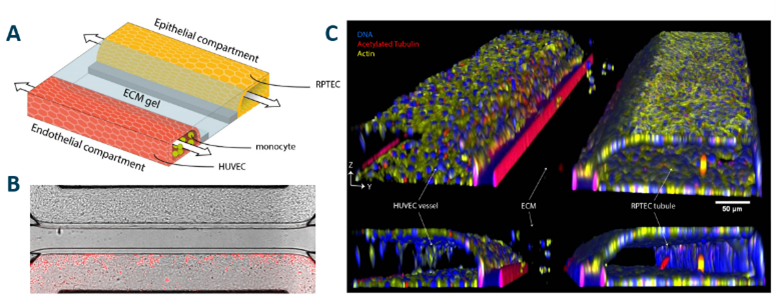
Figure 1. A) 3D schematic of the kidney model, showing RPTEC (yellow), collagen-I ECM gel (blue), HUVECs (red) and monocytes (green). B) Phase contrast image with fluorescent overlay of the model on day 6, showing labeled monocytes in red. C) 3D reconstruction stained for acetylated tubulin (red), actin (yellow) and nuclei (blue).
Modeling and Assessing Kidney Inflammation
Complement activation as well as recruitment and infiltration of immune cells such as monocytes are key steps of an inflammatory response in the proximal tubule which can lead to significant damage. To introduce immune components, fluorescently labeled primary human monocytes are perfused through the endothelial channel. Kidney inflammation was induced using complement activated serum (CAS) added to the renal compartment. This resulted in morphological changes of the epithelium: (A), increased expression of ICAM-1 in the epithelium (B) and increased IL-6 release (C).
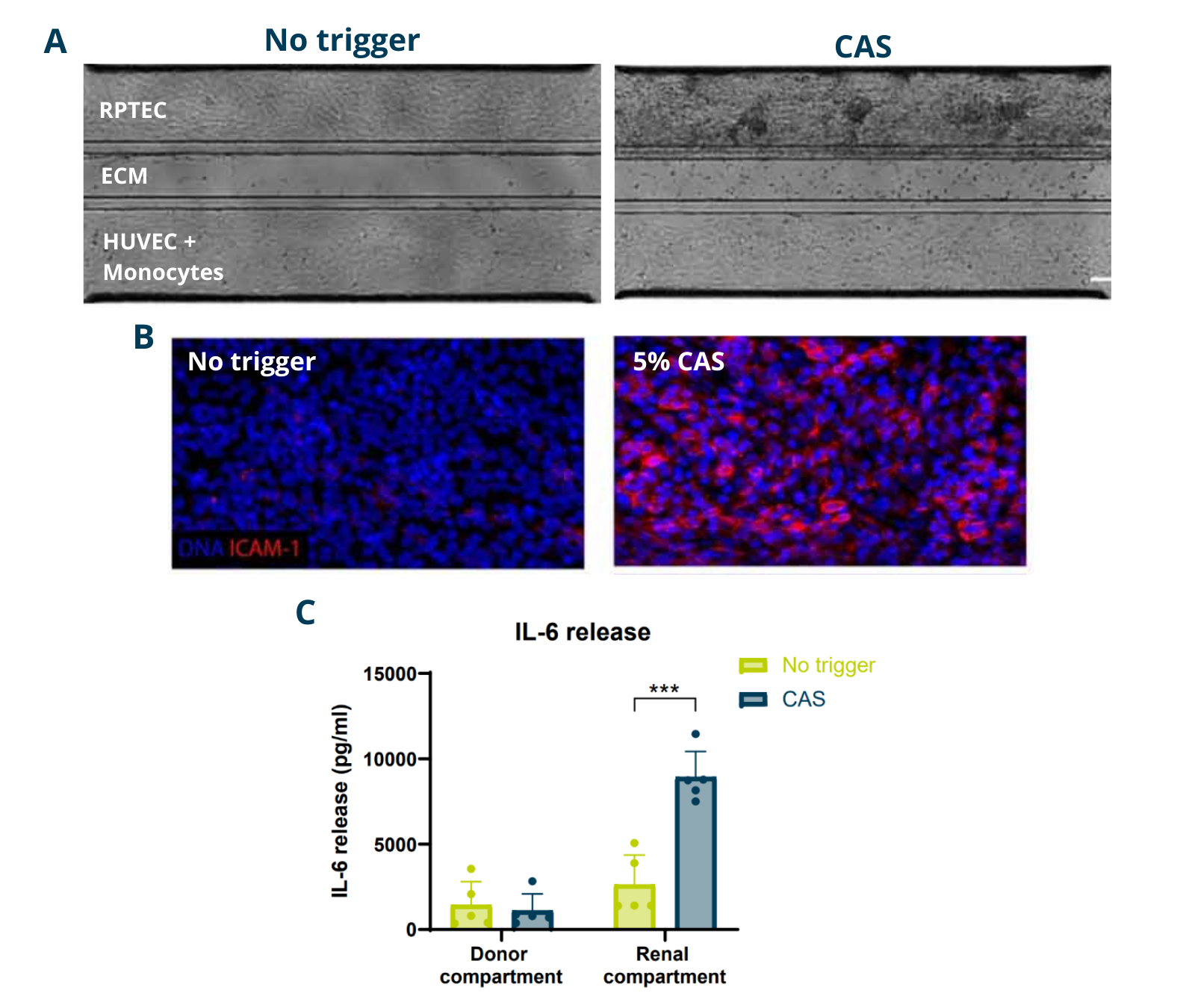
Figure 2. Analysis of the proximal tubule–blood vessel co-culture model after 96 h with or without 5% CAS exposure. (A) Phase contrast images showing monocyte migration into the ECM and proximal tubule. (B) DNA (blue) and ICAM-1 (red) staining of the renal compartment (maximum intensity projections). (C) IL-6 secretion in donor and renal compartments.
Donor-Specific Responses
To assess the model’s reliability for drug development across multiple donors, primary human monocytes from four donors were used in the tri-culture model and challenged with CAS. Results revealed migration of immune cells in response to complement triggered inflammation across donors (D).
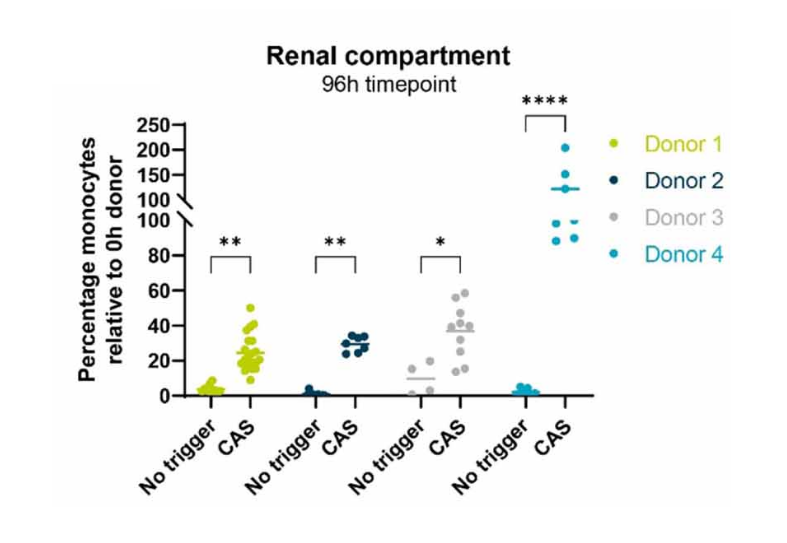
Figure 3. A) Percentage of monocytes present in the Renal compartment after exposure to no trigger or CAS for 4 different donors.
Evaluating Therapeutic Interventions
The model was used for ranking compounds targeting kidney inflammation. Two immune-modulating compounds were evaluated: Compound A, targeting the inflammatory trigger and Compound B, directly targeting monocyte activity. Both compounds significantly reduced monocyte migration into the renal compartment, demonstrating the system’s applicability for screening and evaluating therapeutic strategies aimed at preventing immune-mediated kidney injury.
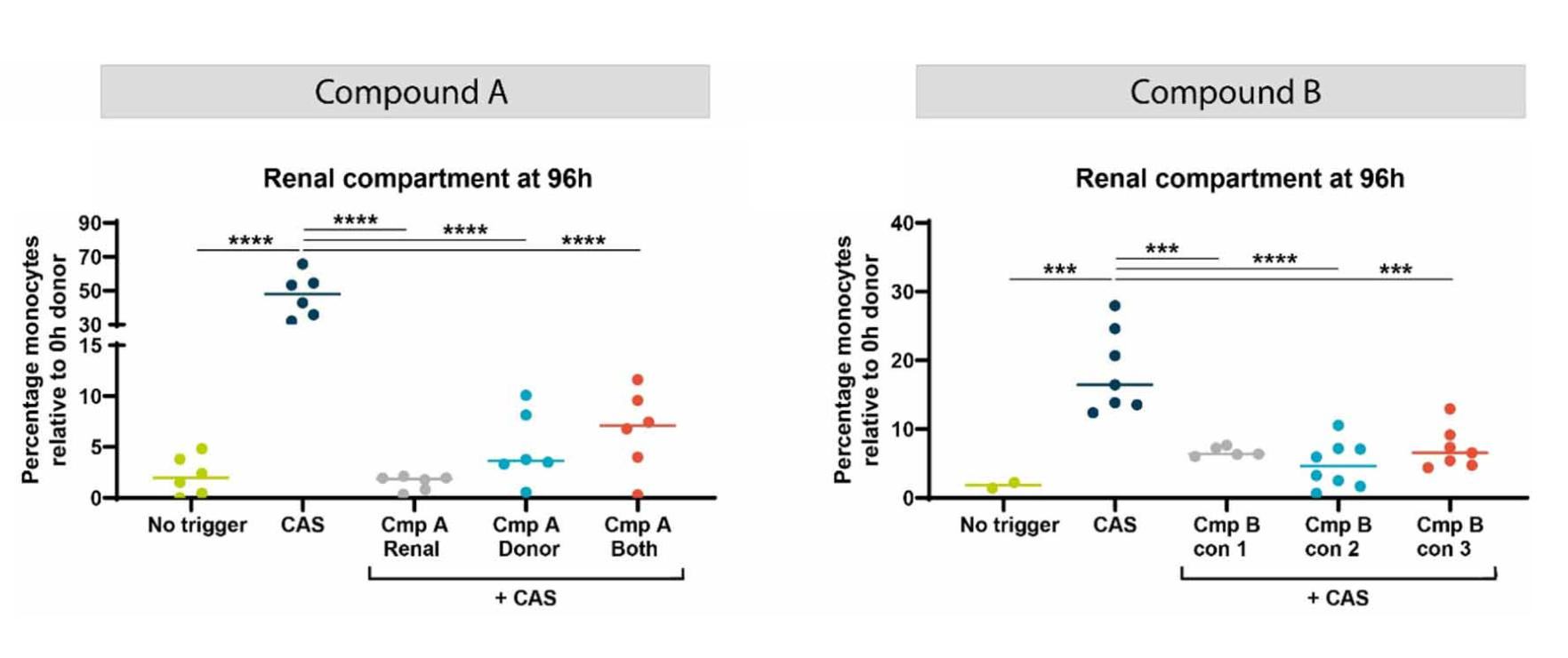
Figure 4. Percentage of monocytes present in the Renal compartment after exposure CAS + compound A or B respectively after 96h. Compound A targets the inflammatory trigger and Compound B targets the monocytes directly. Different exposure strategies were applied based on compound characteristics.
Summary
• Immune competent proximal tubule model was deployed that is comprised of renal epithelium, a blood vessel, ECM, and immune cells. Endothelium and epithelium is exposed to shear stress, which is an important cue for increased polarization, differentiation and transporter functionality.
• Multi donor Inflammation response: by incorporating monocytes and complement activated serum of different donors. Monocytes migrated in response to inflammatory trigger across donors.
• Modulating inflammation through orthogonal mechanisms. Inflammation was reversed by antibodies targeting complement or immune migration, both leading to decreased inflammatory state of the proximal tubule.
• Versatile Platform for Drug Discovery. This high-throughput, immunocompetent human kidney tri-culture model offers a promising preclinical platform to study kidney function and inflammation in both fundamental research and drug screening.
Selected Resources
.avif)

.svg)


.avif)

