Vascular Inflammation-on-a-Chip: A Scalable Platform for Trans-endothelial Electrical Resistance and Immune Cell Migration
Leiden, January 30, 2023 – MIMETAS scientists develop a vasculature inflammation model using a scalable and automation-compatible platform and present a method to study vascular barrier integrity in real-time.
The vasculature system plays an important role in the acute inflammatory process of the body. Certain chronic diseases and autoimmune disorders can induce an aberrant inflammatory response which can become harmful to the body. Part of this chronic inflammation is considered endothelial dysfunction and is characterized by increased vessel permeability and endothelial activation. To screen targets and develop drugs to treat chronic vascular inflammation, it is critical to create models that recapitulate changes in vascular barrier permeability in inflammatory conditions as well as develop methods to accurately quantify the model’s barrier integrity.
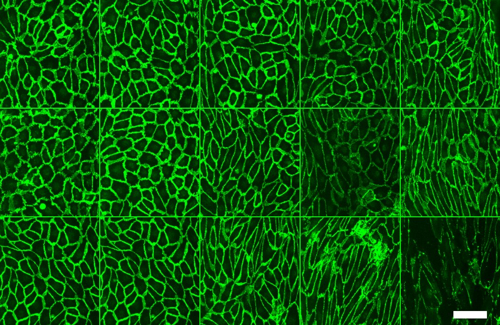
Gaps within conventional in vivo and cell culture vascular models have spurred the development of 3D in vitro vascular barrier function model using microfluidic organ-on-a-chip technologies such as the OrganoPlate®. Robust models have allowed chemical, mechanical, and biological integral to vasculature to be incorporated. In this study, MIMETAS scientists developed a scalable and real-time method to study vascular barrier integrity by combining the speed and sensitivity of TEER based measurements with the automation-compatible OrganoPlate 3-lane 64. The scientists developed 64 microfluidic Human umbilical vein endothelial cells (HUVEC) tubules (OrganoReady® Blood Vessel HUVEC) and induced inflammatory conditions by adding inflammatory cytokines or human peripheral blood mononuclear cells. Using the OrganoPlate 3-lane 64-compatible TEER device, they were able simultaneously evaluate barrier integrity of the HUVEC tubules in real-time under perfused conditions and sensitively detect low barrier resistance. They were also able to mimic the changes of barrier using just cytokines that we also observed using PBMCs. They then multiplexed the assay to add in immunofluorescent staining to look at ICAM and VE-cadherin to highlight changes in cell morphology.
The results showed a significant dose- dependent effect on endothelial barrier of different inflammatory cytokines triggers over a 48-hour time period. Ultimately, in addition to successfully modeling vascular inflammation, the results presented here also highlight the potential for the OrganoTEER and OrganoPlate 3-lane 64 in enhancing models for the blood brain barrier, immuno-oncology, and other diseases.
